There is a lot of conflicting information about fluoroquinolone antibiotics (Cipro, Levaquin, Avelox, Floxin and a few others) noted in scientific journals. One study will conclude one thing about how fluoroquinolones effect human cells and another study will reach the opposite conclusion. It’s frustrating for everyone involved and it leads to the conclusion, that is also noted in most journal articles about fluoroquinolones, that, “Despite their widespread application, the exact mechanism of action of the quinolones is not fully understood.” (1) Despite the fact that the exact mechanism of action of fluoroquinolones is unknown, shouldn’t some of the details of their effects on human cells be known? Shouldn’t there be some clarity in how these drugs affect cells, if not how they work or sometimes don’t work? Basic, verifiable, answers are sought, but they remain elusive. Some interesting, and possibly useful, information may be found in examining why clear answers are so difficult to obtain.
Shouldn’t it be testable whether fluoroquinolones increase or decrease levels of Reactive Oxygen Species (ROS)? Shouldn’t the question of whether fluoroquinolones increase or decrease cellular inflammation be verifiable? Shouldn’t Scientists know whether fluoroquinolones activate or inhibit t-cell gene expression? These things can be studied in laboratories. Answering these questions doesn’t require long-term studies, surveys that are subject to interpretation or vague definitions. They should be answerable questions and the answers should be clear. It’s science, not philosophy. The answers should be black or white, yes or no, not shades of grey.
Yet with each of these questions there are multiple conflicting reports. No one seems to be able to consistently verify what happens to human cells when they are exposed to fluoroquinolones. Some studies done by well-run institutions and published in reputable journals say that fluoroquinolones decrease ROS, reduce inflammation and inhibit t-cell gene expression (2). Other articles in equally well-respected journals say the opposite (3, 4, 5, 6). So which is true? Does the arrow go up or down? I’m sure that answering these questions isn’t easy, but they should be answerable and the answers should be the same each time an experiment is done, right?
So why are there differing answers? Why can’t Scientists, many of whom are undoubtedly brilliant and capable, figure this out? A couple of possible answers are that one group of Scientists’ methods are wrong, or that cells react differently to fluoroquinolones with each exposure. Both possibilities are fascinating on some level. If the methodologies of one group of Scientists produce an anti-inflammatory response within cells, but the methodologies of another group of Scientists produce an inflammatory response within cells, perhaps the difference in methodologies holds the key to limiting an inflammatory response in living humans. A cure, or an antidote to the inflammation that is definitely experienced by some people having an adverse reaction to fluoroquinolones, may be revealed from the study methodologies in which an anti-inflammatory response was induced/observed.
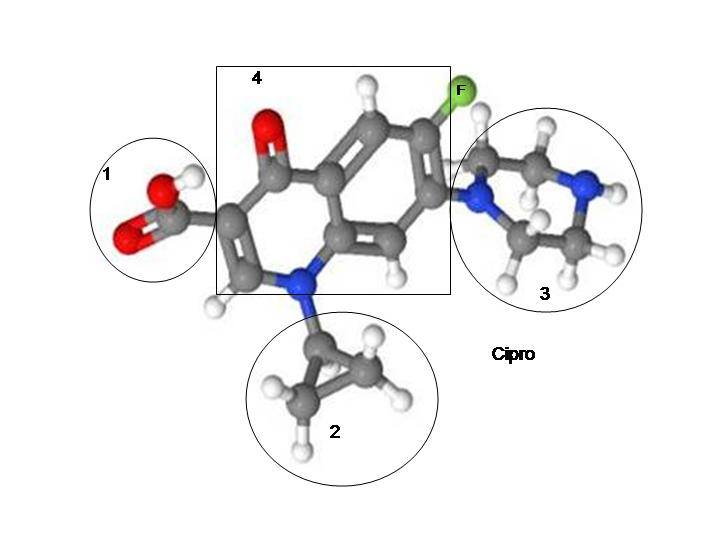
An even more interesting possibility is that how cells react to fluoroquinolones depends on which strand of DNA the quinolone molecules attach to. Studies have found that fluoroquinolones form a poisonous adduct to DNA (7, 8). Perhaps the reaction of the cell in response to exposure to fluoroquinolones depends on which DNA strands are broken, where they’re broken and where the quinolone molecule attaches to the DNA. It is plausible that there are some places where DNA could be broken and adducted to that would create an inflammatory response and there are other places where DNA could be broken and adducted to that would create an anti-inflammatory response. I have neither the tools nor the expertise to test this hypothesis, but from the perspective of someone who has been studying adverse reactions to fluoroquinolones for the past 2 years, the notion that fluoroquinolones break and attach to DNA makes sense of many perplexing aspects about fluoroquinolone toxicity. If we assume that DNA breaks and quinolone adduction to DNA is behind adverse reactions to fluoroquinolones, the following questions may have the following answers:
Why are some people adversely affected by fluoroquinolones while others aren’t? Potential answer – some people have important strands of DNA affected while other people have unimportant strands of DNA affected. And/Or, some people have DNA affected that triggers and inflammatory response and the over-production of ROS, while others don’t because their DNA is broken in less consequential spots.
Why could I handle Cipro for 3 prescriptions but the 4th prescription hurt me? Potential answer – the Cipro affected inconsequential strands of DNA the first 3 times it was administered, but it damaged an important strand of DNA the 4th time it was administered.
Why did I experience a delayed adverse reaction to Levaquin? Potential answer – it takes time for damaged DNA to replicate.
Why can’t anyone seem to figure out how these drugs work? Potential answer – because the human genome is not fully mapped out and most Researchers aren’t looking at how fluoroquinolones affect DNA.
I’m not a Scientist. I certainly could be wrong about the above hypothesis. But I do find it both frustrating and interesting that Scientists, who are undoubtedly smarter than I am, can’t seem to figure out some basic facts about how fluoroquinolones work. I think that there are some answers in their inability to find clear answers. I suspect that the answers lie in quinolone adducts to DNA. Perhaps someone with the tools to determine whether I’m right or wrong will design an experiment (that is consistently verifiable) to determine the effects of fluoroquinolones on DNA, and to determine whether or not DNA damage results in differing effects of the drugs.
Sources:
- Inorganic Chemistry, “New uses for old drugs: attempts to convert quinolone antibacterials into potential anticancer agents containing ruthenium.“
- The Journal of Immunology, “Mitochondrial Reactive Oxygen Species Control T Cell Activation by Regulating IL-2 and IL-4 Expression: Mechanism of Ciprofloxacin Mediated Immunosuppression“
- The Tohoku Journal of Experimental Medicine, “Fluoroquinolone Induced Tendinopathy: Etiology and Preventative Measures“
- Nepal Medical College Journal, “Genotoxic and cytotoxic effects of antibacterial drug, ciprofloxacin, on human lymphocytes in vitro”
- Journal of Young Pharmacists, “Oxidative Stress Induced by Fluoroquinolones on Treatment for Complicated Urinary Tract Infections in Indian Patients“
- Science Translational Medicine, “Bactericidal Antibiotics Induce Mitochondrial Dysfunction and Oxidative Damage in Mammalian Cells“
- The Journal of Biological Chemistry, “The Mechanism of Inhibition of Topoisomerase IV by Quinolone Antibacterials.”
- Proceedings of the National Academy of Sciences of the United States, Biochemistry, “Quinolone Binding to DNA Mediated by Magnesium Ions”











Your questions are very intelligent and noteworthy. Hopefully a scientist or two will pick up on this!
Lisa, you are better than most scientists, because you’re actually asking questions!! Thank you so much!!! It’s such a shame that so few people in the medical industry actually care about looking for answers… or even more importantly, PREVENTING these problems in the first place because they were all trained by the pharmaceutical companies to just push drugs on their patients.
Here’s an interesting piece of the puzzle that might (or might not) have some answers. We know that one of the reasons Cipro is so incredibly dangerous is that it is so high in fluoride, which is one of the most toxic elements on planet earth. So, in essence, floxies have suffered from severe fluoride poisoning.
Looking up fluoride toxicity on toxiperdia.org, it looks like the Cytochrome p450 enzymes within the human body cause defluorination of fluoride within drugs like Cipro. From what I can gather, defluorination is a GOOD THING because it reduces the toxicity of fluoride.
So here’s where it gets interesting… my new doctor who I’ve been seeing since I got floxed specializes in “functional medicine”, which is supposed to be the medicine that looks at the body as a whole. Well, like most doctors, she has turned out to be almost completely worthless because she doesn’t really seem to be that interested in what happened to me, but she IS very interested in selling me $2,000 worth of monthly supplements. However, even though she’s MOSTLY worthless, sometimes she stumbles upon something which proves useful to me later.
She had me do a DNA cheek swab test. It’s called the DetoxiGenomic Profile Test, and it’s from a company called Genova Diagnostics. It tests your genetic profile to see if your particular genes make you potentially susceptible to adverse drug reactions.
My tests came back, and two of my Cytochrome p450 enzymes (CYP2C9 and CYP2C19) have decreased activity, which according to the test results, says that my genes prevent the normal metabolism of drugs, and that severe side effects are possible for me, even at very low dosages. It says this right on my test results… it says that I should use caution when taking prescription drugs.
So, does that mean that my particular Cytochrome p450 enzymes prevented my body from defluorinating the fluoride within Cipro in a timely-enough fashion to get it the hell out of my bloodstream before it destroyed my entire body? Is it possible that certain people’s underactive Cytochrome p450 enzymes make them more susceptible to toxins like fluoride, whereas other people’s genes help them eliminate the toxins immediately… thus preventing those people from getting floxed (or getting more mild versions of getting floxed)??
I don’t really know the answer to this. But my new question is:
If Cipro poisoning is really a super-aggressive and super-advanced form of fluoride poisoning, is there any CURE for fluoride poisoning? I think that more research papers have probably been done on fluoride poisoning than Cipro poisoning, and they’re probably one-and-the-same illness.
Oops… I meant “toxipedia.org”, not “toxiperdia.org”
I’m not sure what is wrong with me I can’t get answers anywhere!!!! I have been suffering for the last 7 or 8 years with pain in my knees, elbows, hips, lower back upper back, bone pain, numbness on the outsides of my thighs, Severe fatigue loss of sleep muscle waisting. I have to make myself go each day I have no choice and the list goes on. I believe it is do to Cipro poisoning, in 2008 I went to my GP and was treated for a UTI and given a weeks worth of Cipro. That is about when all of this started. I have had blood work done over and over again I have been tested for Lyme desease also went to Lyme specialist had other Tick testing done with know answer. I bring up the Cipro poisoning to my GP that prescribed it he says I don’t think so. It took time to finally figure out things and put it together with know help from Doctors. At first I thought it may be cancer because it started under my lower rib cage then from there to other joint tendon tissue. I figure it’s not cancer nothing shows in my blood tests and it’s been going on for almost 8 years now. You don’t only deal with the pain you get stressed and depressed!! If you have any answers about any of this or who I should see it would be much appreciated. I have been waiting for a referral to UVA to see a rhumatologist and try to get answers. I want to say thank you for trying to figure it out and how the poisoning works. Please forgive my spelling errors. Thank you Mike