It has recently been discovered that, “chemicals or genetic mutations that impair topoisomerases, and possibly other components of the transcription elongation machinery that interface with topoisomerases, have the potential to profoundly affect expression of long ASD (autism spectrum disorder) candidate genes.” (1)
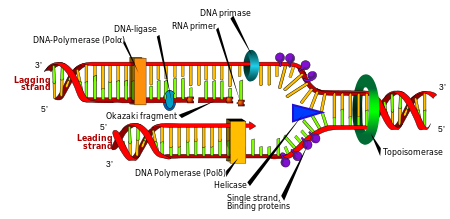
Topoisomerases are enzymes that are essential for DNA and RNA replication and reproduction. Without topoisomerase enzymes, DNA and RNA supercoiling fail to resolve correctly during cell division and gene transcription. Topoisomerases are like oil to the DNA and RNA replication engine—making the process go smoothly. Without topoisomerases, DNA and RNA supercoiling jams up and “over-heats,” causing transcription errors. Topoisomerases are also “integral for gene expression, as they resolve DNA supercoiling that is generated during transcription.” (1)
Many pharmaceutical drugs inhibit topoisomerases. Most of these drugs are chemotherapeutic agents such as topotecan, a drug used to treat various forms of cancer. The most widely used topoisomerase inhibitors are fluoroquinolone antibiotics—Cipro/ciprofloxacin, Levaquin/levofloxacin, Avelox/moxifloxacin and Floxin/ofloxacin—drugs used to treat simple urinary tract, prostate, sinus and other infections. The fluoroquinolone antibiotics that are in wide use were first patented in the late 1980s and their use increased steadily from then until now, with 26.9 million prescriptions for orally and IV administered fluoroquinolones written in 2011. (2) A similar number of topically administered fluoroquinolone prescriptions have been written, and though orally and IV administered fluoroquinolones are not recommended for use in children because they cause cartilage lesions in juvenile animals (3), topically administered fluoroquinolones are approved for use in children as young as 6 months of age (4), and are regularly given to young children in the form of ear and eye drops.
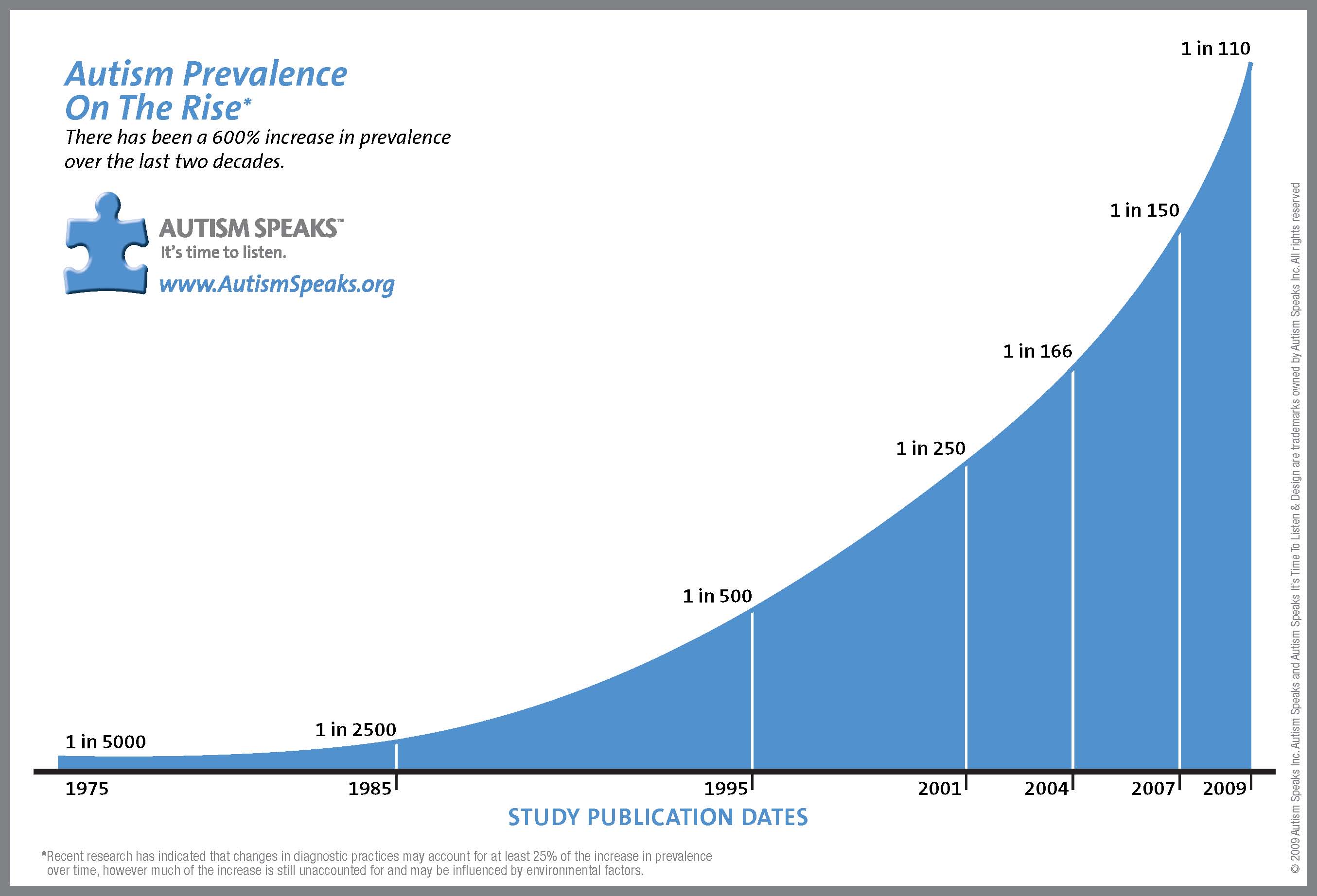
Autism rates have been skyrocketing since the 1980s, with the most recent numbers out of the CDC stating that one in 68 American children is on the autism spectrum. (5)
It is likely that a variety of environmental and genetic components has led to the staggering figure of 1 in 68 American children on the autism spectrum. Studies have focused on vaccines (and there are vaccine injured children), but epidemiological studies have suggested that factors other than vaccines are likely at play. Birth control pills, acetaminophen, antidepressants and other categories of drugs have been pointed to for their deleterious effects on the human brain, and their possible contributions to the increasing number of people on the autism spectrum.
STUDY SUMMARY AND IMPLICATIONS
In this article, I will go over some studies that point to topoisomerase interrupting drugs as a potential cause of autism. The studies are out of the University of North Carolina at Chapel Hill—“Topoisomerases facilitate transcription of long genes linked to autism,” published in Nature (as well as the corresponding author manuscript), and “Topoisomerase 1 inhibition reversibly impairs synaptic function,” published in PNAS. I will link these studies to the fact that fluoroquinolones are also topoisomerase interrupters.
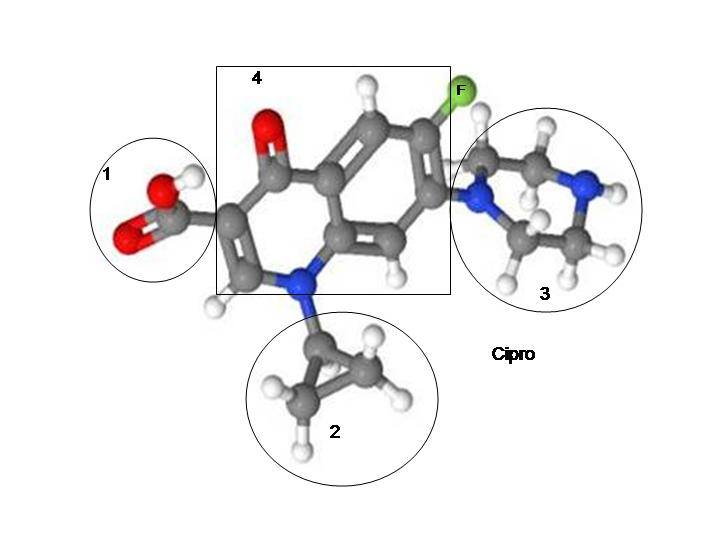
The mechanism of action for Cipro/ciprofloxacin is:
“The bactericidal action of ciprofloxacin results from inhibition of the enzymes topoisomerase II (DNA gyrase) and topoisomerase IV (both Type II topoisomerases), which are required for bacterial DNA replication, transcription, repair, and recombination.”(6)
The mechanism of action for other fluoroquinolones—Levaquin/levofloxacin, Avelox/moxifloxacin, Floxin/ofloxacin, and their generic equivalents—are the same.
Why anyone thought that it was a good idea to give DNA disrupting chemotherapeutic drugs to children with ear infections is beyond my comprehension, but it happens every day.
Fluoroquinolone use has gone up hand in hand with autism rates. As critics of all possible causes of autism are quick to point out, correlation does not mean causation. While that criticism is true, the causes of autism are undoubtedly correlated with autism rates, and thus correlations should be examined.
The articles that I will be reviewing note that topoisomerase interrupting drugs profoundly affect the expression of genes related to autism. A large number of the genes that have been found to be related to autism are particularly long and complex, and are related to neurotransmitter synaptic function and neurodevelopment. The expression of these long genes is affected by topoisomerase interrupting drugs, as one may expect when noting that topoisomerases are necessary for gene transcription, and longer genes can more easily get mis-transcribed when exposed to topoisomerase interrupting drugs.
Topoisomerase enzymes are expressed throughout the adult brain, and thus the connections between topoisomerase inhibiting drugs and neurodegenerative diseases should be explored along with connections between those drugs and autism. It is noted in “Topoisomerases facilitate transcription of long genes linked to autism” that, “Our findings suggest that chemicals and genetic mutations that impair topoisomerases could commonly contribute to ASD and other neurodevelopmental disorders.”
Both “Topoisomerases facilitate transcription of long genes linked to autism” and “Topoisomerase 1 inhibition reversibly impairs synaptic function” examine the effects of topotecan, a topoisomerase 1 (TOP1) inhibitor, on autism related genes and synapses. Studies of the effects of fluoroquinolones, topoisomerase 2 and 4 interrupters, have not yet been published.
It should be noted that the existing studies have looked at nuclear gene expression, and that the effects of topoisomerase interrupting drugs on the expression of mitochondrial and microbiome genes have not yet been explored.
The implications of “Topoisomerases facilitate transcription of long genes linked to autism” and “Topoisomerase 1 inhibition reversibly impairs synaptic function” are potentially huge given the wide-spread use of topoisomerase interrupting fluoroquinolones, both directly in humans and in agriculture. If fluoroquinolones are conclusively linked to autism, their use in children and people of child-bearing age (exactly how gene expression is affected by topoisomerase interrupting drugs, and what the intergenerational effects may be, are not yet known) should be prohibited—effective immediately—regardless of what the average physician who knows little about the effects of topoisomerase inhibitors on gene expression thinks about the “excellent safety record” of fluoroquinolones as a class of drugs.
As a person who has been hurt by ciprofloxacin, a fluoroquinolone antibiotic, I am not without bias—but I do not think that it’s unreasonable to assert that all topoisomerase interrupting drugs should be strictly limited—especially if they’re connected in any way to autism or neurodegenerative diseases.
STUDIES LINKING TOPOISOMERASE INTERRUPTING DRUGS TO AUTISM
“Topoisomerases facilitate transcription of long genes linked to autism” concludes that:
“Our data suggest that chemicals or genetic mutations that impair topoisomerases, and possibly other components of the transcription elongation machinery that interface with topoisomerases, have the potential to profoundly affect expression of long ASD candidate genes. Length-dependent impairment of gene transcription, particularly in neurons and during critical periods of brain development, may thus represent a unifying cause of pathology in many individuals with ASD and other neurodevelopmental disorders.”
This conclusion has multiple levels of significance. It involves a shift in thinking about ASD as either a genetic disorder or an environmentally caused disorder, to noting the interplay between genes and the environment (epigenetics).
The study is also significant in that it notes that genes that encode synaptic function and neurodevelopment, that are also related to autism, are particularly long and complex. Those long and complex genes aren’t transcribed properly when topoisomerase enzymes are inhibited via pharmaceuticals. When topoisomerase interrupting drugs impair gene transcription, expressions of long genes become impaired.
Whether or not the silencing of expression of particularly long genes through topoisomerase interrupting drugs is a possible unifying cause of autism spectrum disorders depends on the prevalence of topoisomerase interrupting drugs in our environment. Topotecan, the topoisomerase 1 interrupter that was studied, is a chemotherapeutic drug that is only used in cancer patients, and is rarely used in pediatric patients. Fluoroquinolone antibiotics, on the other hand, are topoisomerase interrupting drugs that are used commonly in the general population, including the pediatric population, and are even used in agriculture and thus are present in the food we eat, the soil our food is grown in and even the water we drink.
More than 20 million prescriptions for fluoroquinolones—topoisomerase interrupting drugs—have been written each year for more than two decades. One is hard pressed to find an adult American who has not had at least one fluoroquinolone prescription in his or her lifetime. Fluoroquinolone use has gone up hand in hand with incidence rates of autism. As noted earlier, that correlation does not mean causation, but if topoisomerase interrupting drugs in our pharmacies and environment are a unifying cause of ASD (and other neurodegenerative diseases of modernity), fluoroquinolones would need to be the causal factor, not prudently used chemo drugs like topotecan. Studies looking into this line of thinking are pending, and the similarities between fluorquinolones and chemotherapeutic topoisomerase interrupters have not escaped the attention of the researchers looking into the relationships between topoisomerases and autism.
The UNC researchers found that, “topotecan reduced expression of nearly all extremely long genes in mouse cortical neurons,” (1) and were able to reproduce the same results in human neurons. Interestingly, not only was the expression of long genes suppressed, “topotecan increased expression of a majority of genes that were <67 kb in length, although the magnitude of this increase was very small for most genes.” (1) The expression of long genes, those genes that are involved in encoding neural synapses, was downgraded, whereas the expression of shorter genes was up-regulated.
An example of a particularly long gene that is related to ASD whose expression is altered by topotecan is “Ubiquitin-protein ligase E3A (Ube3a), a gene that affects synaptic activity and that is deleted or duplicated in distinct neurodevelopmental disorders (Angelman syndrome and autism, respectively)” (7) Ube3a is “normally expressed only from the maternal allele in neurons and regulates synaptic function.” (1) However, in cells that have been exposed to topoisomerase interrupting drugs, the paternal allele of Ube3a is transcriptionally upregulated. “Duplication of the chromosomal region containing maternal Ube3a is frequently detected in individuals with autism.”
Other particularly long genes are affected by topoisomerase interrupting drugs include:
“many genes down-regulated by topotecan are associated with synapses, cell adhesion, and neurotransmission. Moreover, a number of those down-regulated long genes are associated with autism, including Neurexin-1 (Nrxn1; 1059 kb), Neuroligin-1 (Nlgn1; 900 kb) and Contactin-associated protein 2 (Cntnap2; 2,241 kb), genes that are well known to regulate inhibitory and excitatory synaptic function.” (7)
I wasn’t able to find any scholarly articles about the effects of fluoroquinolones on Nrxn1, Nlgn1 or Cntnap2. However, it is hypothesized in “Epigenetic side-effects of common pharmaceuticals: A potential new field in medicine and pharmacology,” that all adverse reactions to fluoroquinolones are due to epigenetic mechanisms:
“The quinolones are a family of broad-spectrum antibiotics. They inhibit the bacterial DNA gyrase or the topoisomerase IV enzyme, thereby inhibiting DNA replication and transcription. Eukaryotic cells do not contain DNA gyrase or topoisomerase IV, so it has been assumed that quinolones and fluoroquinolones have no effect on human cells, but they have been shown to inhibit eukaryotic DNA polymerase alpha and beta, and terminal deoxynucleotidyl transferase, affect cell cycle progression and function of lymphocytes in vitro, and cause other genotoxic effects. These agents have been associated with a diverse array of side-effects including hypoglycemia, hyperglycemia, dysglycemia, QTc prolongation, torsades des pointes, seizures, phototoxicity, tendon rupture, and pseudomembranous colitis. Cases of persistent neuropathy resulting in paresthesias, hypoaesthesias, dysesthesias, and weakness are quite common. Even more common are ruptures of the shoulder, hand, Achilles, or other tendons that require surgical repair or result in prolonged disability. Interestingly, extensive changes in gene expression were found in articular cartilage of rats receiving the quinolone antibacterial agent ofloxacin, suggesting a potential epigenetic mechanism for the arthropathy caused by these agents. It has also been documented that the incidence of hepatic and dysrhythmic cardiovascular events following use of fluoroquinolones is increased compared to controls, suggesting the possibility of persistent gene expression changes in the liver and heart.”(8)
Also, it has been known since 1996, when “Delayed Cytotoxicity and Cleavage of Mitochondrial DNA in Ciprofloxacin-Treated Mammalian Cells” was published in Molecular Pharmacology, that fluoroquinolones deplete mitochondrial DNA. The article states, “The loss in mtDNA was associated with a delayed loss in mitochondrial function. Here, we report that the 4-quinolone drug ciprofloxacin is cytotoxic to a variety of cultured mammalian cell lines at concentrations that deplete cells of mtDNA.” (9) Nuclear gene expression is linked to mitochondrial functioning and, “Mitochondria generate signals that regulate nuclear gene expression via retrograde signaling,” (10). Fluoroquinolone induced mitochondrial DNA damage may lead to changes in nuclear DNA expression.
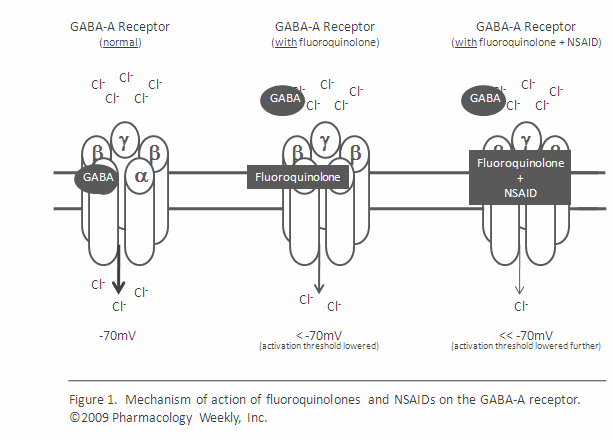
Fluoroquinolones are also known to affect neurotransmitters, particularly GABA neurotransmitters. (11) GABA neurotransmitters are responsible for regulation of inhibitory and excitatory synaptic function. It is noted in “Topoisomerase 1 inhibition reversibly impairs synaptic function” that, “GABA-A receptor subunits are encoded by long genes.” And that, “multiple synaptic proteins encoded by long genes including cell adhesion molecules linked to autism and GABA receptor subunits, are depleted in topotecan-treated neurons.”
Given that GABA receptors are critical for inhibitory and excitatory synaptic functioning, examining the role between GABA receptors, topoisomerases, topoisomerase inhibiting drugs, and attention deficit hyperactivity disorder (ADHD) is certainly warranted. Many individuals with ASD also display symptoms of ADHD, and the interactions between GABA and glutamate receptors may be why many individuals with ASD find symptom relief through a gluten and casein free diet that is low in glutamate.
As the title of “Topoisomerase 1 inhibition reversibly impairs synaptic function” indicates, it was found that “the synaptic effects of topotecan are reversible” upon washout of the drug. However, the complexity of neurotransmission is acknowledged in the discussion section of the article, where it is noted that, “adding back one synaptic cell-adhesion molecule would not likely restore the protein levels of all affected synaptic cell-adhesion molecules as well as multiple GABA-A receptor subunits.” It is also noted that, “transient TOP1 (topoisomerase 1) inhibition has the potential to impair brain function reversibly, whereas a persistent change in TOP1 activity has the potential to disrupt neurodevelopment and promote neurodegeneration.” With limited exposure to a topoisomerase interrupting drug, the neurological effects appear to be reversible. However, persistent exposure can cause more persistent harm.
Adult patients with fluoroquinolone toxicity syndrome often note that they were able to tolerate fluoroquinolones without experiencing adverse effects prior to getting “floxed.” This tolerance threshold may indicate that fluoroquinolones somehow accumulate in cells and that the adverse effects of fluoroquinolones are amplified with each exposure. It is also possible that mitochondrial DNA needs to cross over a damage threshold before resulting in adverse effects. More about this can be found in the post, “The Fluoroquinolone Time Bomb – Answers in the Mitochondria.”
IS IT POSSIBLE THAT FLUOROQUINOLONE ANTIBIOTICS ARE RESPONSIBLE FOR MANY CASES OF AUTISM?
The possibility hasn’t been explored, despite the documented effects of fluoroquinolones that line up with many of the symptoms and effects of autism. In addition to the correlation between fluoroquinolone prescription rates and autism rates, and the points noted above about topoisomerase interrupting drugs changing the expression of autism related genes, fluoroquinolones also damage mitochondria (12)—and mitochondrial damage has been linked to autism (13) . Fluoroquinolones also cause cellular leakage (14) and depletion of minerals (15) that are necessary for synthesis of minerals and vitamins that (deficiencies of) have been linked to autism (16). Fluoroquinolones, as powerful antibiotics, are extremely destructive to the gut microbiome and gut microbiome health has also been linked to autism (17).
Even though a pretty good argument can be made for fluoroquinolones causing autism, if there is a relationship, it is not entirely clear-cut. There are children who have been hurt by fluoroquinolones, but there are also children who have been administered fluoroquinolones without any apparent harm. Like adults, children almost certainly have a tolerance threshold for fluoroquinolone use before they are injured. Delayed adverse reactions (18) make recognition of symptoms of fluoroquinolone toxicity difficult to recognize, and recognition in the pediatric population is even more difficult because children have incompletely developed communication skills. Studies that take into account delayed reactions and tolerance thresholds for fluoroquinolones have not been conducted on the adult population, much less the pediatric population, and thus it is unknown what the true effects of fluoroquinolones are.
It is possible that parental genes are altered by fluoroquinolone use and the damaged genes are passed on to children. This possibility has not yet been explored, but, anecdotally, it does not appear to be a clear-cut relationship either. Many mothers and fathers who have suffered from fluoroquinolone toxicity have had neurodevelopmentally normal children. Some have had neurodevelopmentally challenged children though too, and it would be nice to see an actual study of the children of parents exposed to fluoroquinolones, as opposed to the anecdotes that are currently available.
Autism is a complicated disorder (or set of disorders) that likely has multiple environmental, genetic and epigenetic causes. I strongly suspect that fluoroquinolones are part of the autism-cause-puzzle, but I also suspect that other cellular poisons compound the deleterious effects of fluoroquinolones on neural synapses, and can trigger autism.
I noted above that adverse reactions to fluoroquinolones are often delayed. Delayed adverse reactions to fluoroquinolones have been reportedly triggered by other stressors, including other pharmaceuticals (especially NSAIDs and steroids), benzodiazepine withdrawal, vigorous exercise, emotional stress, alcohol, hormonal changes and other things that can alter neurotransmitters and the autonomic nervous system. The combined effects of fluoroquinolones and subsequent stressors have not yet been studied, despite (some) recognition of delayed adverse reactions to fluoroquinolones.
Given the indications of “Topoisomerases facilitate transcription of long genes linked to autism,” and “Topoisomerase 1 inhibition reversibly impairs synaptic function,” it is certainly prudent to explore whether or not fluoroquinolones affect gene expression similarly to topotecan. Other drugs that inhibit topoisomerase activity, such as irinotecan and camptothecin, demonstrated similar effects to those of topotecan and thus the UNC researchers “speculate that other drugs that inhibit TOP1 or TOP2 enzymes could have similar effects on synaptic function.” We shall see if their results show that fluoroquinolones affect the expression of long genes. I’m betting that they will find that fluoroquinolones dramatically alter gene expression, especially after multiple uses and also especially when combined with NSAIDs. Given the prevalence of both fluoroquinolone antibiotics and NSAIDs in our environment though, I hope that I am wrong.
It was never prudent for fluoroquinolones to be used as first-line antibiotics. They are dangerous, DNA and RNA disrupting chemo drugs that should only be used in life-or-death situations, not as treatment for travelers’ diarrhea or sinus infections. They’re topoisomerase interrupters. They’re topoisomerase interrupters. They’re topoisomerase interrupters. They’re topoisomerase interrupters. I’m not sure how many times I need to say that for people who know what topoisomerases are (those who went to medical school) to recognize that it is NOT APPROPRIATE TO GIVE TOPOISOMERASE INTERRUPTING DRUGS TO CHILDREN (or anyone else who isn’t dying).
A former biochemist friend noted, “I was stunned that people thought quinolones were perfectly safe. Coming through the ranks, I always thought, as was taught, that they were a last-ditch drug. There was absolutely no long-term research done with them, and apparently still isn’t. But there sure was money involved.”
Long-term and intergenerational research need to be done on drugs that affect DNA before they are released into the public. Neither long-term nor intergenerational studies were done on fluoroquinolones before flooding the market with them. The odd intricacies of how fluoroquinolones affect people were not taken into consideration either—things like delayed effects, tolerance thresholds, drug and hormonal interactions, etc.
I’m hopeful that the UNC scientists that are looking at the interactions between topoisomerases and autism will start screaming about the deleterious effects of fluoroquinolones on our genes. I hope that they have the resources to do the study with delayed adverse reactions and tolerance effects taken into consideration too.
We shall see.
I’m hopeful that the study will be well-done and, if not conclusive, interesting–and that it opens the door for more research.
I can’t say that I hope that fluoroquinolones are a unifying cause of autism spectrum disorders, because that would be too sad.
One thing that I’ve found through researching how fluoroquinolones damage human health, is an appreciation for how complex and multifaceted humans are. Nothing happens in isolation. There are feedback and feedforward loops within the body that compound the effects of a stimulus on one bodily system on another, there are interactions between drugs that can occur long after a drug “should” have cleared the body, the interactions between mitochondria and neurotransmitters and cellular signaling exist but little is known about them, and more. It is difficult, if not impossible, for scientists to adequately take into account the incredible complexity of the human body. What is not known cannot be taken into consideration, and don’t for a second think that we know “enough” about the human body to be throwing pharmaceuticals that are topoisomerase interrupters into the mix.
Fluoroquinolones never should have been approved for human use until long-term, intergenerational safety studies were performed. They should be taken off of the market until those studies are done. That won’t happen though. Drugs aren’t taken off of the market unless they immediately kill a lot of people. The damage that fluoroquinolones do is more nuanced than that. If pharmaceutical makers can create a drug that is complex enough, and that can affect people in multiple different ways—through affecting gene expression—they can get away with just about anything. Bayer and Johnson & Johnson have figured this out. The FDA isn’t smart enough to protect the public. Individuals need to be smart enough to protect themselves and their families—a difficult thing to do considering that some knowledge of biochemistry and genetics is necessary for protection against dangerous pharmaceuticals.
Fluoroquinolones are topoisomerase interrupters. They’re topoisomerase interrupters. They’re topoisomerase interrupters. They’re topoisomerase interrupters. They’re topoisomerase interrupters. Repeat until someone who should know better shouts – “STOP.”
REMAINING QUESTIONS
The existing research into connections between topoisomerase interrupting drugs and autism spectrum disorders raises multiple questions. It would be nice if some money, time, effort and other resources were devoted to answering these questions.
- What are the effects of fluoroquinolone antibiotics on nuclear, mitochondrial and microbiome gene expression? What are the implications of these effects?
- Do topoisomerase interrupting drugs change gene expression of the person who takes them, the offspring of the person who takes them, or both?
- Do topoisomerase interrupting drugs increase a person’s chances of having a child with Autism? How?
- If a person takes a topoisomerase interrupting drug, is their DNA altered? If so, are the changes temporary or permanent?
- Are some people’s genes affected by these drugs more than others? What factors determine whether or not an individual’s genes are affected?
- Are DNA/gene alterations triggered by pharmaceuticals reversible? If so, how?
- What, if anything, can people who have taken these drugs do to discourage the expression of the ASD related genes?
- When would the administration of the drug happen to influence genes in a way that could trigger the genes associated with Autism – when a mother is pregnant or at any point before the child is conceived – or does the drug need to be directly administered to the person whose genes are altered?
- Do these drugs change gene expression in the ways that diet and music change gene expression or do they change DNA like Agent Orange? What level and scale are we talking about?
SOURCES:
- Ian F. King, et al. “Topoisomerases facilitate transcription of long genes linked to autism” Nature – author manuscript. 2013 Sep 5; 501(7465): 58–62.
- FDA Safety Announcement, “FDA Drug Safety Communication: FDA requires label changes to warn of risk for possibly permanent nerve damage from antibacterial fluoroquinolone drugs taken by mouth or by injection” 08/15/2013
- Adam D. “Use of quinolones in pediatric patients.” Reviews of Infectious Diseases. 1989 Jul-Aug;11 Suppl 5:S1113-6.
- Ciprodex Warning Label
- CDC Newsroom Press Release, “CDC estimates 1 in 68 children has been identified with autism spectrum disorder” March 27, 2014
- FDA Warning Label for Cipro/Ciprofloxacin
- Mabb AM, et al. “Topoisomerase 1 inhibition reversibly impairs synaptic function.” Proceedings of the National Academy of Sciences of the United States of America, 2014 Dec 2;111(48):17290-5. doi: 10.1073/pnas.1413204111. Epub 2014 Nov 17.
- Antonei B. Csoka and Moshe Szyf. “Epigenetic side-effects of common pharmaceuticals: A potential new field in medicine and pharmacology.” Medical Hypotheses 73 (2009) 770–780
- JW Lawrence, et al. “Delayed cytotoxicity and cleavage of mitochondrial DNA in ciprofloxacin-treated mammalian cells.” Molecular Pharmacology November 1996 vol. 50 no. 5 1178-1188
- Martin Pickard, et al. “Progressive increase in mtDNA 3243A>G heteroplasmy causes abrupt transcriptional reprogramming” Proceedings of the National Academy of Sciences of the United States of America, 2014 Sep 23; 111(38): E4033–E4042.
- Pharmacology Weekly Newsletter, “What is the mechanism by which the fluoroquinolone antibiotics (e.g., ciprofloxacin, gemifloxacin, levofloxacin, moxifloxacin) can increase a patient’s risk for developing a seizure or worsen epilepsy?” August 31, 2009.
- Sameer Kalghatgi et al. “Bactericidal Antibiotics Induce Mitochondrial Dysfunction and Oxidative Damage in Mammalian Cells.” Science Translational Medicine, 5, 192ra85 (2013)
- Suzanne Goh, et al. “Mitochondrial Dysfunction as a Neurobiological Subtype of Autism Spectrum Disorder – Evidence From Brain Imaging” JAMA Psychiatry. 2014;71(6):665-671
- Marta Kicia, et al. “Comparison of the effects of subinhibitory concentrations of ciprofloxacin and colistin on the morphology of cardiolipin domains in Escherichia coli membranes.” Journal of Medicinal Microbiology, April 2012 vol. 61 no. Pt 4 520-524
- G Palu, et al. “Quinolone binding to DNA is mediated by magnesium ions” Proceedings of the National Academy of Sciences of the United States of America, Vol. 89, pp. 9671-9675, October 1992
- Richard E. Frye, MD, PhD, and Daniel A. Rossignol, MD, FAAFP, “Cerebral Folate Deficiency in Autism Spectrum Disorders” Autism Science Digest: The Journal of Autism One. Issue 02.
- Jennifer G. Mulle, et al. “The Gut Microbiome: A New Frontier in Autism Research.” Current Psychiatry Rep. 2013 Feb; 15(2): 337.
- Jacquelyn K. Francis, BA and Elizabeth Higgins, MD, “Permanent Peripheral Neuropathy: A Case Report on a Rare but Serious Debilitating Side-Effect of Fluoroquinolone Administration” Journal of Investigative Medicine High Impact Case Reports 1–4 © 2014 American Federation for Medical Research
- Thomas D. Gootz, et al. “Inhibitory Effects of Quinolone Antibacterial Agents on Eucaryotic Topoisomerases and Related Test Systems” Antimicrobial Agents and Chemotherapy. Jan. 1990, P. 8-12.
- Mukherjee, S. Sen, K. Agarwal, “Ciprofloxacin: mammalian DNA topoisomerase type II poison in vivo.” Mutation Research Letters. Volume 301, Issue 2, February 1993, Pages 87–92













I thought this site was one of hope. Just more negative news for all to think about. I think you should rethink your focus.
Because the topoisomerases damage is caused by gene constitutive mutation, the gene mutation is an effective measure to solve this problem.